The general formula is that the nth shell can in principle hold up to 2 n 2 electrons. The 1 st innermost shell s, can hold up to two electrons, the 2 nd shell can hold up to eight (2s 2 + 2p 6) electrons, the 3 rd shell can hold up to 18 (3s 2 + 3p 6 + 3d 10) electrons. These in turn are divided into s, p, d, and f orbital types 4 (sometimes called subshells) each capable of holding a maximum of 2 electrons. Shells, also called principal energy levels, are designated K, L, M or 1, 2, 3. The structure of an atom consists of protons and neutrons in the nucleus, and electrons that move around the nucleus in specific energy levels, known as shells. 
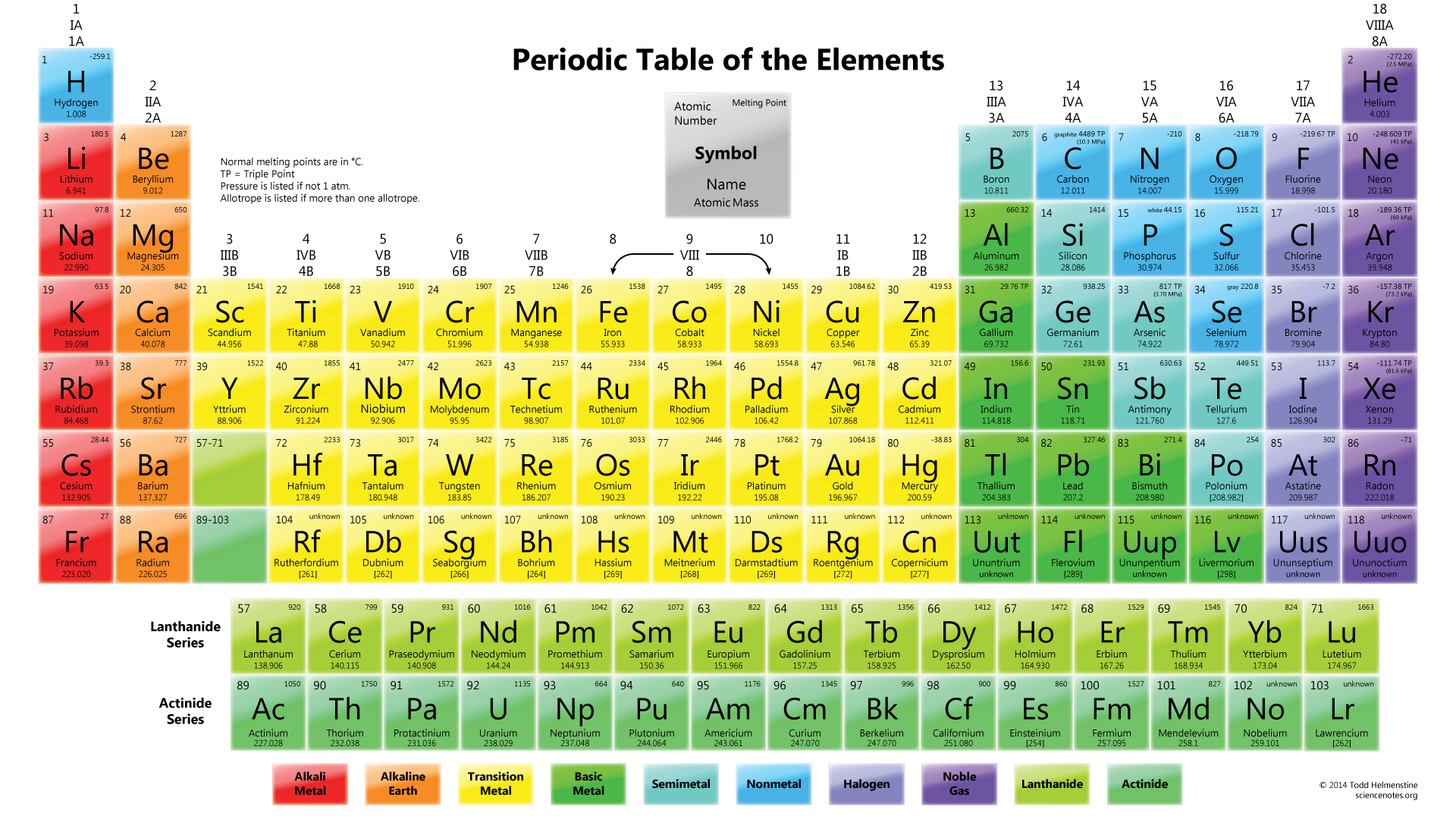
This material, otherwise known as an element, cannot be broken down or changed into another substance by chemical means. An atom is the smallest unit of matter that has all the chemical properties of that particular substance. Figure 1.Ĭomprehension of the Periodic Table’s organization may seem like a daunting task but a fundamental understanding can be achieved after a description of atomic structure. The atomic weight is the total weight of protons plus neutrons found in the nucleus. Most prominent is atom type, distinctively identified by a two letter symbol, its etymology primarily from abbreviations of Latin and Greek names, or the discoverer of the element, and the atomic number of an element Z 3 which is equal to the number of protons in the nucleus. 
Chemists use this table in their work, and everyone can understand structural beauty from a creator God. Atoms are periodic because of the way that subatomic particles are arranged and added to each atom hence they can be systematically placed in a table. Atoms’ organized structure leads to predictable attributes and chemical behaviour, so predictions can be made on how atoms will bond or react with other atoms to form complex, multi-atom chemical compounds such as DNA. Order and pattern can be clearly seen in the table, one of the most powerful and perhaps least understood tools of science. When the new element was discovered and named ‘germanium’, its properties closely matched Mendeleev’s predictions of ‘ekasilicon’ for atomic mass, density, and melting point. Mendeleev predicted the discovery of a missing element he called ‘ekasilicon’ on paper after observing a gap in the Periodic Table between silicon and tin. There were spaces where elements could be located but no one at that time had discovered them. Mendeleev saw that the 65 then-known elements in the table lie at the heart of chemistry but that it was incomplete. Awakening, I immediately wrote it down on a piece of paper-only in one place did a correction later seem necessary.” 2 Mendeleev’s table is noteworthy because it exhibits the most accurate values for atomic mass allowing one to recognize trends over the entire array of elements. 1 “I saw in a dream a table where all elements fell into place as required. Mendeleev predicted the discovery of a missing element he called ‘ekasilicon’ on paper after observing a gap in the Periodic Table between silicon and tinĭmitri Mendeleev (1834–1907), a Russian chemist, developed a formal organization of the elements in 1869 after having a dream. When God created, He brought order to the universe even in the smallest things, for God is not the author of confusion (1 Corinthians 14:33). Looking into the nature of atoms, creation is clearly seen. This orderliness is not a random or haphazard assemblage of particles happening by accident or spontaneously organizing without an intelligent cause. Order starts with atoms and the subatomic particles that comprise them. Everything we observe from plant life, stars, animals, rocks, air, and water-virtually everything-is composed of 90 naturally occurring building blocks known as atoms. The beauty and organization of God’s creation can be seen all around us in the macroscopic world. Perman Dream idea: Dmitri Mendeleev (1834–1907). Atoms and God’s order in the fundamental building blocks of all substance The Periodic Table of chemical elementsīy Craig A. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
